New Carbohydrate Oxidase-cellulose Binding Domain Fusions for Enzyme Immobilization in Textile Bleaching and Laundry Innovation Cases.
The OXIPRO team at the Universitat Autònoma de Barcelona (UAB) have identified a carbohydrate binding module in the CBM3 family as a very good candidate for improving the immobilization properties of NAG oxidase when fused together in the OXIPRO Textile Innovation Case.
Collaborative work between UAB and Novozymes has resulted in the successful fusion of CBM3, a bacterial domain, with a fungal carbohydrate oxidase (already used in the Textile Innovation Case). A related oxidase has been benchmarked for hydrogen peroxide generation in OXIPRO WP5 (Smart integration of enzyme innovations in a consumer product relevant environment) for the Detergent Innovation Case (An-Glucose Oxidase). It is desirable for the carbohydrate oxidase-binding domain fusions to be tried in both the OXIPRO Textile and Laundry Innovation Cases.
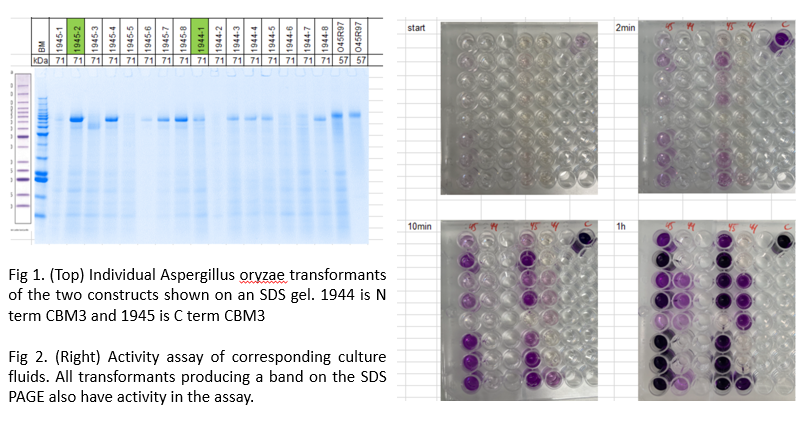
Results for expression of the fusions in Aspergillus oryzae so far show that the carbohydrate binding module CBM3 can be fused to either the N terminal or C terminal configuration on the oxidase and that both appear active in a model oxidase assay with glucose as substrate.

Fig 1. Individual Aspergillus oryzae transformants of the two constructs shown on an SDS gel. 1944 is N term CBM3 and 1945 is C term CBM3

Fig 2. Activity assay of corresponding culture fluids. All transformants producing a band on the SDS PAGE also have activity in the assay.
Enzymes are being purified at Novozymes before shipment to UAB and LEITAT for testing. The incorporation of these purified carbohydrate binding modules could allow for immobilization of the oxidase in the textile bleaching process (Textile Case) and for enhancement of the performance of peroxide generation close to the fabric during a laundry cycle (Detergent Case).